By: Andrew Yoon, M.D., medical director, Heart Failure, MemorialCare Heart & Vascular Institute at Long Beach Medical Center.
MemorialCare Heart & Vascular Institute at Long Beach Medical Center is one of only two medical centers in California selected to participate in the RESPONDER-HF confirmatory trial, a global clinical study evaluating the Corvia® Atrial Shunt. This first-of-its-kind device is designed to treat heart failure with preserved ejection fraction (HFpEF), often referred to as “heffpeff” heart failure—a condition characterized by sudden, debilitating shortness of breath with minimal physical activity.
HFpEF is among the most challenging forms of heart failure to manage. Many patients continue to struggle despite optimal medical therapy, making everyday activities feel insurmountable. The Corvia Atrial Shunt offers a targeted, device-based solution opening blood flow in the heart to give patients back their ability to breathe, which once felt insurmountable for heff-peff patients.
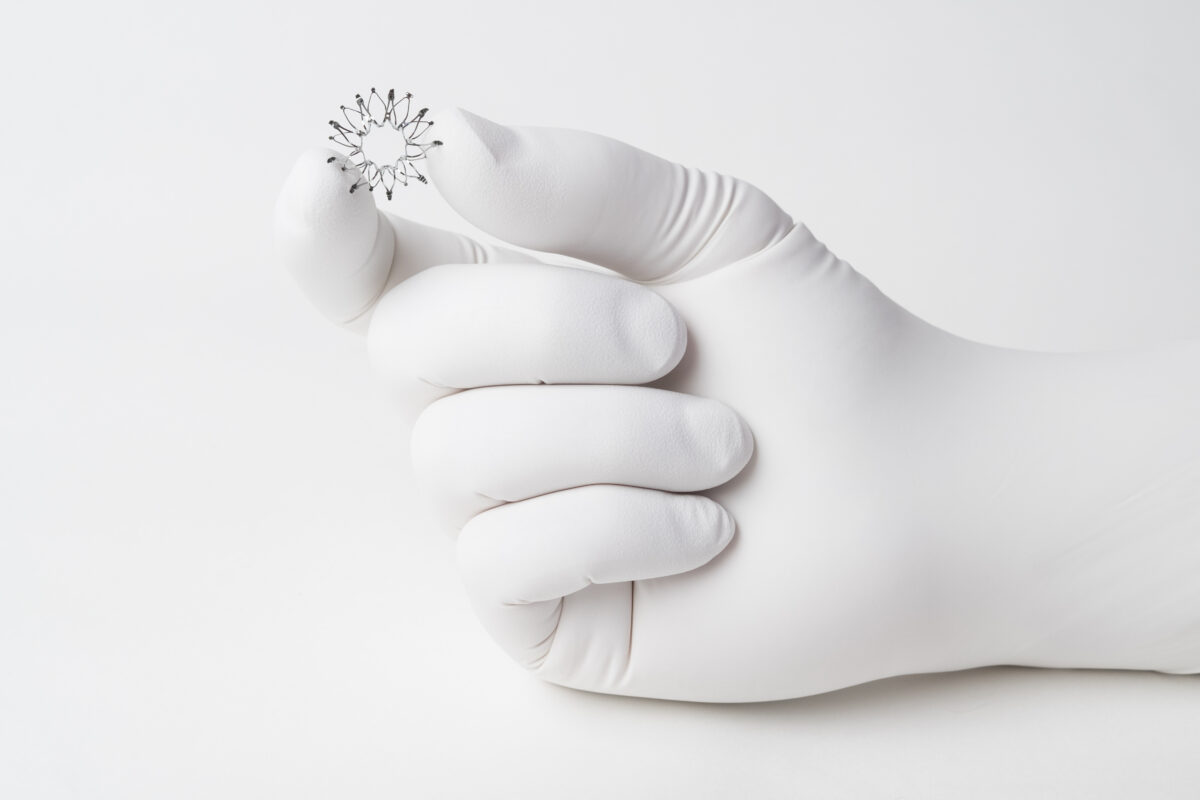
The Corvia Atrial Shunt works by creating a controlled passage between the heart’s two upper chambers, the left and right atria. Implanted through the femoral vein using a minimally invasive approach, the dime sized device is designed to reduce procedural risk and shorten recovery time. Once in place, the shunt allows blood to flow from the left atrium to the right atrium during physical activity, when pressure in the left atrium rises abnormally. At rest, when pressures are normal, there is minimal or no blood flow across the device. This dynamic response helps decompress the heart and lungs during exertion, allowing patients to feel relief from shortness of breath more quickly.
By relieving elevated left atrial pressure, the Corvia Atrial Shunt targets hallmark HFpEF symptoms such as breathlessness during activity, fatigue, and fluid retention. These symptoms are among the most common reasons patients with HFpEF seek emergency care or experience repeat hospitalizations. Clinical studies have demonstrated sustained improvements in quality of life, exercise capacity, and symptom relief, with no significant device-related complications reported.
HFpEF also disproportionately affects women. Of the estimated 13 million people worldwide diagnosed with the condition, approximately 60% are women. Heart failure in women is frequently underdiagnosed or undertreated due to delayed recognition, atypical symptom presentation, and disparities in access to specialized care. Expanding access to innovative therapies such as the Corvia Atrial Shunt represents an important step toward addressing these gaps and improving outcomes for women with heart failure.
Participation in this landmark trial reflects Long Beach Medical Center’s ongoing commitment to advancing cardiovascular care. Recently designated as an Extracorporeal Cardiopulmonary Resuscitation (ECPR) Receiving Center—one of only five hospitals in Los Angeles County to achieve this distinction—the medical center continues to serve as a regional leader in lifesaving cardiac interventions. With expertise in minimally invasive valve surgery, transcatheter therapies, ECMO support, and comprehensive heart failure management, MemorialCare Heart & Vascular Institute works closely with referring providers throughout Southern California to deliver innovative, individualized care and help shape the future of heart failure treatment.
Learn more about the RESPONDER-HF trial at the MemorialCare Heart & Vascular Institute at Long Beach Medical Center by calling (657) 241-9052, or visiting memorialcare.org/heart.